First trial phase to take place at Memorial Sloan Kettering Cancer Center
Pictured: Samples under microscope used to identify cancer. PHOTO CREDIT: uri.edu
Professors at the University of Rhode Island are working in collaboration with Yale University and Memorial Sloan Kettering Cancer Center (MSKCC) on a new peptide that has been designed to target cancerous tumors and make them more recognizable for surgeons to remove or to be targeted for diagnostic or therapeutic interventions.
The peptide, named pHLIP®, stands for pH-Low Insertion Peptide, and was developed with collective efforts from professors from URI and Yale beginning in 2004. Yana Reshetnyak and Oleg Andreev are physics professors at URI who work together with Donald Engelman, Professor of Molecular Biophysics and Biochemistry at Yale.
According to an article published by URI News, the pHLIP molecule was originally discovered (in Engelman’s Lab) from a protein called Bacteriorhodopsin, which is composed of multiple helix formations bound together. One of the helices in Bacteriorhodopsin was observed to be able to pass across cell membranes if the membrane was acidic or had a low pH
“‘When pHLIP encounters a cell membrane with a neutral pH, it will sit on the surface briefly and then pull away,’” said Engelman in another article from URI News. “‘But if it’s in an acidic environment, then the peptide folds into a helix, crosses the cell membrane and stays there.’”
Yana Reshetnyak and Oleg Andreev, two URI physics professors, knew that cancer cells tend to have a lower pH of 5.5 – 6.5 compared to healthy cells ranging around 7.4., and realized that pHLIP could be used for specific targeting of acidic tumors and delivery of imaging and therapeutic payloads to tumors. Because of the lower pH found in cancer cells, the pHLIP molecule is able to single out their attachments to only cancer cells and avoid attachment to normal cells all due to their pH.
Reshetnyak, Engelman and Andreev and their Labs have been working together for over 20 years to investigate molecular mechanisms of pHLIP peptides and their applications in medicine.
“I think I always wanted maybe to do something in this field that would help people to survive this very horrible disease,” Andreev said.
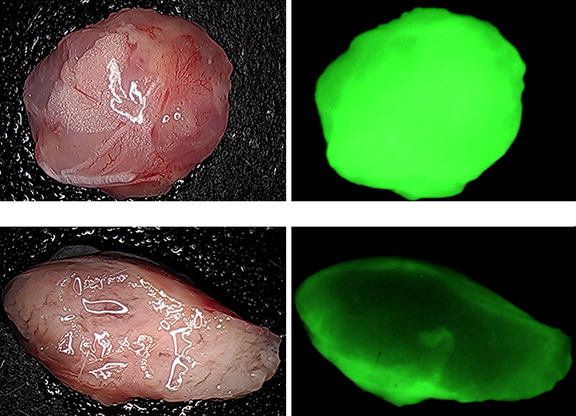
pHLIP is being combined with different molecules to use their ability of accurate attachment to cancer cells for multiple research projects and different clinical applications. One study is using pHLIP along with a fluorescent dye called ICG. With pHLIP being a transporter for ICG, the molecule is able to target tumor cells making them glow to produce a fluorescent light. Surgeons are able to remove the glowing tumors, making surgery more accurate at removing cancerous lesions and ensuring clean margins.
“pHLIP ICG is basically the idea that you can sort of apply a filter and do it with near infrared fluorescence, and it will glow,” said Hannah Visca, a physics Ph.D. student working in Andreev’s Lab. “If we can get that real time feedback through fluorescence, thats a huge help.”
Another project where pHLIP is being utilized is with the addition of a STING agonist (STimulator of InterferoN Gene). Activation of STING protein is able to make tumors that are disguised with cancer cells more noticeable to the body’s immune system. The STING proteins activated by STING agonists release a protein called Interferon, which alerts the immune system of a foreign cell. pHLIP is able to transport STING agonists specifically to tumor cells with much more accuracy compared to non-targeted uses of STING agonists.
Both projects have seen improved success when pHLIP molecules have been used as a targeted delivery. According to an article posted by URI News, pHLIP-ICG experiments using the removed bladders in bladder cancer patients have seen an improvement in detection by 17%. This combined molecule has proved enough success to have their first phase of a surgically-driven clinical trial take place at the Memorial Sloan Kettering Cancer Center for breast cancer patients.
There have also been promising results with pHLIP-STING agonist research. A singular injection of the pHLIP-STING agonist was administered into 20 mice that had cancerous tumors. Eighteen of those mice saw complete elimination of their tumors within a few days of receiving the injection. Moreover, the immune memory was developed: re-injection of cancer cells did not lead to tumor growth. However, 10 mice received an injection of just the STING agonist molecule, and all tumors remained in the mice with only some minor reduction in growth.
Micheal Dupont is a PhD student at URI and collaborated on the pHLIP-STING project with Professor Andreev for around three years. He said that having the ability to work on a project that helps surgeons operate with accuracy as well as create a successful treatment is very important to him in earning his PhD.
Although the pHLIP molecule has been recognized for its advancement, there is still a lot more work to be done to transfer it to clinics, according to Reshetnyak. In the next three years, she hopes their patented molecule will be translated to multiple clinical trials and will be tested for use in clinics for cancer imaging and treatment.
Reshetnyak and her colleagues from Yale and MSKCC founded pHLIP, Inc company to develop clinics and commercialize pHLIP technology. More information about pHLIP technology and its mechanism of action could be found on a company website phlipinc.com